SNOWTEX ® is colloidal silica with many applications for paper, fibers, steel and catalysts. Most recently, it has been used by the semiconductor industry as a polishing agent for silicon wafers. Without the development of chemicals such as SNOWTEX, the electronics industry would not be where it is today.
Only Nissan Chemical produces proprietary particle shapes such as SNOWTEX-UP and SNOWTEX-PS.
SNOWTEX comes in a variety of types to suit your needs.
Applications of SNOWTEX
SNOWTEX has a wide variety of industrial applications such as:
- Catalyst binder
- Adhesion and coating
- Binding and hardening
- Polishing
- Investment casting
- Impregnation filler
- Battery manufacturing
- Modification and strengthening of organic resins
Particles and Surface State
SNOWTEX® is colloidal silica made by growing mono-dispersed, negatively charged, amorphous silica particles in water. OH ions exist at the surface of the particles with an electric double layer formed by alkali ions. Stabilization is achieved by the repulsion between the same negatively charged particles. Disturbance of the charge balance will cause the colloidal silica to aggregate, resulting in high viscosity or gelation of the solution.
Stability
Effects of pH change
The higher the concentration or the smaller the particle size, the greater the effect pH change will have on gelation time and stability. SNOWTEX is very stable in the 2-4 and 8-10 pH ranges as illustrated in the diagram below. If a formulation or coating system has a neutral pH, or the formulation requires a colloidal silica with greater stability, SNOWTEX-C can be used.
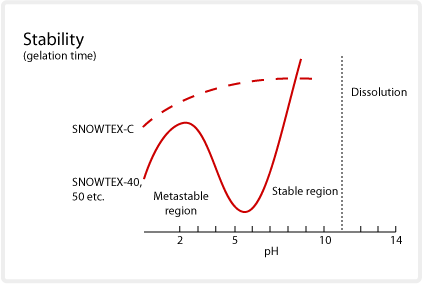
When the pH exceeds 10.5 with a strong alkali, such as caustic soda or sodium silicate, coagulation and dissolution of the silica particles occurs.
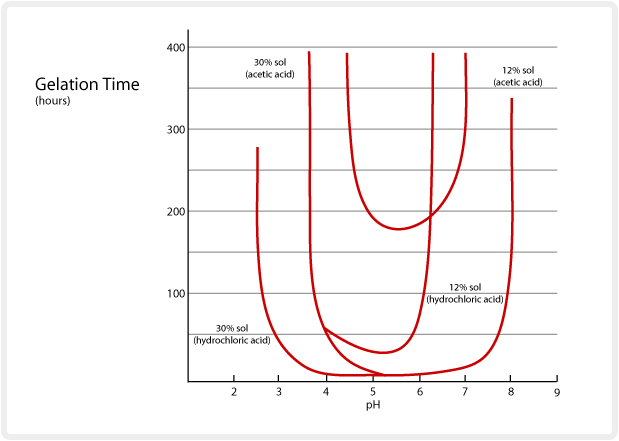
Long chain amines tend to cause SNOWTEX to gel, but ethylamine, morpholine and other similar amines tend to stabilize the sol. SNOWTEX maintains its stability for a fairly long time even when the pH of an alkaline SNOWTEX grade is lowered by the addition of an acid. However, the gelation time differs with the kind of acids used for pH adjustment. The gelation time when the pH is changed by adding hydrochloric acid and acetic acid is shown below. Organic acids tend to provide better stability in terms of gelation time, depending on the SiO2 concentration and particle size.
Effects of salts and electrolytes
Polyvalent metal ions will cause SNOWTEX to gelate. This is because the polyvalent ions interfer with the surface charge of the SNOWTEX. The resulting charge imbalance causes the particles to agglomerate. The surface charge maintains the SNOWTEX stability. The degree of gelation is not uniform, and varies depending on the type, concentration, temperature, etc. of the electrolyte used. Monovalent salts react slowly, but polyvalent salts such as alum, react quickly at pH 9 and higher. When the pH is lowered, the effect of alum and magnesium sulfate decreases. This electrolytic effect on SNOWTEX is useful to speed up or slow down the gelation. The gelation time of SNOWTEX at 52° C with addition of sodium chloride and sulfate are depicted below.
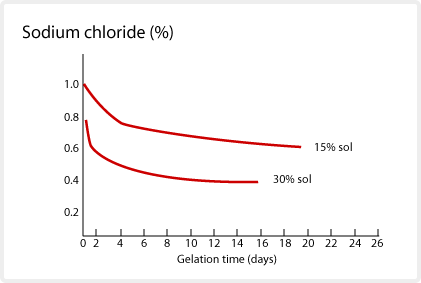
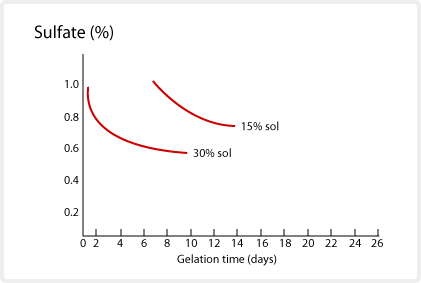
Compatibility with organic solvents
Alcohol, acetone and other polarized solvents can be mixed with SNOWTEX. However, there is a mixing limit and gelation may occur. The compatibility with alcohol when the pH is changed is shown below as an example. SNOWTEX-O can be mixed with alcohol at any proportion.
When alcohol is mixed with SNOWTEX-20 the stability is very good. *
| SNOWTEX-20 (grams) | METHANOL (grams) | ETHANOL (grams) | Stability (85°C) | |
|---|---|---|---|---|
| No.1 | 90 | 10 | – | 24 hours or more |
| No.2 | 80 | 20 | – | 24 hours or more |
| No.3 | 90 | – | 10 | 24 hours or more |
| No.4 | 80 | – | 20 | Gelled in 13 hours |
|
*This will dilute SNOWTEX-20. SNOWTEX-20 contains 20% SiO2 by weight.
|
||||
Lowering the pH of the SNOWTEX-20 with hydrochloric acid to 3.9 and then mixing the alcohol significantly reduces the stability.
| SNOWTEX-20 (grams) | METHANOL (grams) | ETHANOL (grams) | Stability (85°C) | |
|---|---|---|---|---|
| No.5 | 90 | 10 | – | 1 hour 10 minutes |
| No.6 | 80 | 20 | – | 1 hour 45 minutes |
| No.7 | 90 | – | 10 | 1 hour 28 minutes |
| No.8 | 80 | – | 20 | 1 hour 50 minutes |
Compatibility with surfactants
Generally, SNOWTEX is compatible with anionic and nonionic surfactants. Compatibility is dependent on impurities and the composition of the surfactant. Incompatibility may cause the SNOWTEX to gel or agglomerate. Therefore, it is important to select the surfactant carefully. SNOWTEX-C is compatible with a wide range of surfactants.
Compatibility with emulsion resins and water soluble resins
SNOWTEX is compatible with emulsion resins and water soluble resins having the same pH and electric charge. In the case of emulsion resin, gelation and separation may occur depending on which emulsifier is used. Therefore, it is important to select the emulsifier carefully. SNOWTEX-C is also compatible with a wide range of emulsifiers.
Dry Gel
When SNOWTEX is dried, a dry gel is formed. Slowing the drying time will produce smaller agglomerates as well as greater particle-size distribution. This results in a harder dry gel solid.
Thermal change
The illustration below compares the differential thermal analysis (DTA) and the thermal gravimetric analysis of SNOWTEX dry gel. The transition of the silica particles is highlighted in the illustration below.
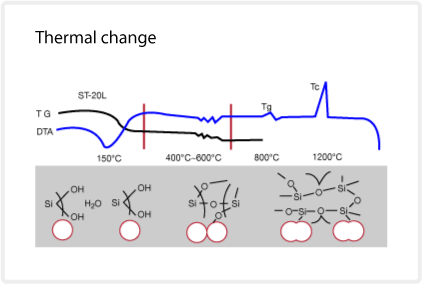
At approximately 150°C, water desorbs from the silica gel, resulting in a weight reduction of approximately 5%. Dehydration of the silanol groups at 400-700°C causes another small weight reduction. Up to this temperature, the particles remain unchanged, but at approximately 800°C, surface fusion between the silica particles occurs. At the glass transition (Tg), the mechanical strength of the silica increases. At approximately 1200°C , an exothermic peak is caused by crystallization (Tc) into cristobalite. The melting point is estimated at 1700°C. The glass transition temperature, crystallization temperature and melting point depend on the amount of sodium ion in the SNOWTEX. The melting points of SNOWTEX-O and SNOWTEX-N are higher because they contain less sodium ions.
| SNOWTEX ® | ST-20L | ST-O | ST-OL | ST-ZL |
|---|---|---|---|---|
| SiO2 (wt%) | 20-21 | 20-21 | 20-21 | 40-41 |
| Na2O (wt%) | < 0.3 *2 | < 0.04 | < 0.05 | < 0.07 |
| pH | 9.5-11.0 | 2-4 | 2-4 | 9.0-10.0 |
| Particle Shape | Spherical | Spherical | Spherical | Spherical |
| Particle Size (nm) | 40-50 | 10-20 | 40-50 | 70-100 |
| Viscosity (mPa.s. at 25°C) | < 3 | < 3 | < 3 | < 5 |
| Specific Gravity (25°C) | 1.12-1.14 | 1.12-1.14 | 1.12-1.14 | 1.29-1.32 |
| Appearance | Opalescent | Clear to opalescent | Opalescent | Opalescent |
|
*1: Flammable alkali (as Na2O) is less than 0.04 wt%. And NH3 is less than 0.2 wt%.
*2: Flammable alkali (as Na2O) is less than 0.03 wt%. |
||||
Note: These are typical properties of SNOWTEX.
Properties of SNOWTEX-PS
The unique string-of-pearl particles and high viscosity of SNOWTEX-PS give it excellent bonding and coating properties. That, in conjunction with its high fluidity, make SNOWTEX-PS excellent for:
- Coating paper, plastics and metals
- Binder for catalyst
- Modification of paints and coating agents
- Fiber treatment
- Binding agent
- Casting
| SNOWTEX ® | ST-PS-M | ST-PS-S | ST-PS-SO |
|---|---|---|---|
| SiO2 (wt%) | < 0.2 | 15-16 | 15-16 |
| Na2O (wt%) | < 0.20 | < 0.02 | |
| pH | 9.0-10.5 | 9.0 – 10.5 | 2-4 |
| Particle Shape | String-of-pearls | String-of-pearls | String-of-pearls |
| Particle Size (nm) / Dynamic light scattering method |
18-25/80-150 | 10-15/80-120 | 10-15/80-120 |
| Viscosity (mPa.s. at 25°C) | < 100 | < 100 | < 100 |
| Specific Gravity (25°C) | 1.12-1.14 | 1.08 – 1.11 | 1.08 – 1.11 |
| Appearance | Milky white | Milky white | Milky white |
|
String-of-pearl particles have a diameter of 18-25 nm with a length of 80-100 nm.
|
|||
Note: These are typical properties of SNOWTEX.
Properties of SNOWTEX-UP
The colloidal particle of SNOWTEX-UP has a unique, elongated shape unlike typical, spherical silicasol. SNOWTEX-UP has many advantages.
The elongated particle shape and high viscosity of SNOWTEX-UP give it excellent properties for film forming, bonding and less migration.
This can be useful for:
- Coating paper, plastics and metals
- Binder for refractories
- Binder for ceramics, glass and fiber
- Textile treatment
| SNOWTEX ® | ST-OUP | ST-UP |
|---|---|---|
| SiO2 (wt%) | 15-16 | 20-21 |
| Na2O (wt%) | < 0.03 | < 0.02 |
| pH | 2-4 | 9.0-10.5 |
| Particle Shape | Elongated | Elongated |
| Particle Size (nm) | 9-15/40-100 | 9-15/40-100 |
| Viscosity (mPa.s. at 25°C) | < 20 | < 100 |
| Specific Gravity (25°C) | 1.08-1.11 | 1.12-1.14 |
| Appearance | Opalescent | Opalescent |
|
Elongated particles have a diameter of 9-15 nm with a length of 40-100 nm.
Ammonia-stabilized ST-UP is available upon request. |
||
Note: These are typical properties of SNOWTEX.
